Item CL002002: As a jar of water is warmed from the bottom, the water at the bottom of the jar becomes warmer than the water above it and starts to rise, and its thermal energy rises with it.
A jar of cold water is placed over a flame and the flame warms the water at the bottom of the jar, increasing its thermal energy.
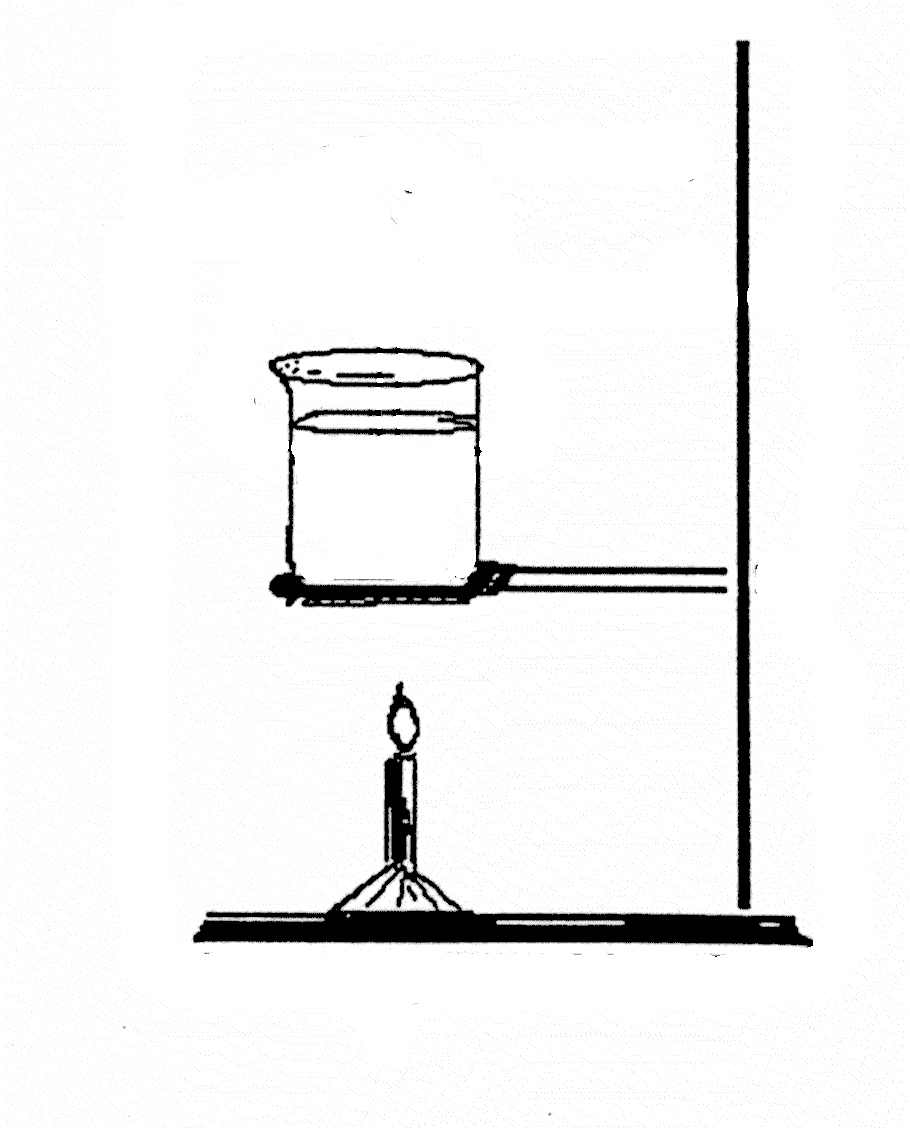
What happens to the thermal energy of the water at the bottom of the jar?
- As the water at the bottom of the jar gets warmer than the water above it, the water rises and its thermal energy rises with it.
- Nothing moves until the water starts to boil, and then the water at the bottom of the jar rises and its thermal energy rises with it.
- The thermal energy of the water at the bottom of the jar rises but the water itself does not rise.
- The thermal energy of the water at the bottom of the jar rises and the water rises too, but the water and the thermal energy rise separately.
- Distribution of Responses

- Students Responding Correctly
| Group | Correct | Total | Percent |
|---|---|---|---|
| Overall | 956 | 2251 | 43% |
| Grades | |||
| 6–8 | 468 | 1197 | 39% |
| 9–12 | 486 | 1043 | 47% |
| Gender | |||
| Male | 463 | 1089 | 43% |
| Female | 467 | 1070 | 44% |
| Primary Language | |||
| English | 849 | 1921 | 44% |
| Other | 74 | 235 | 31% |

